North Carolina-Based LabCorp Receives Emergency Clearance By FDA For COVID-19 Self-Collection Test Kit
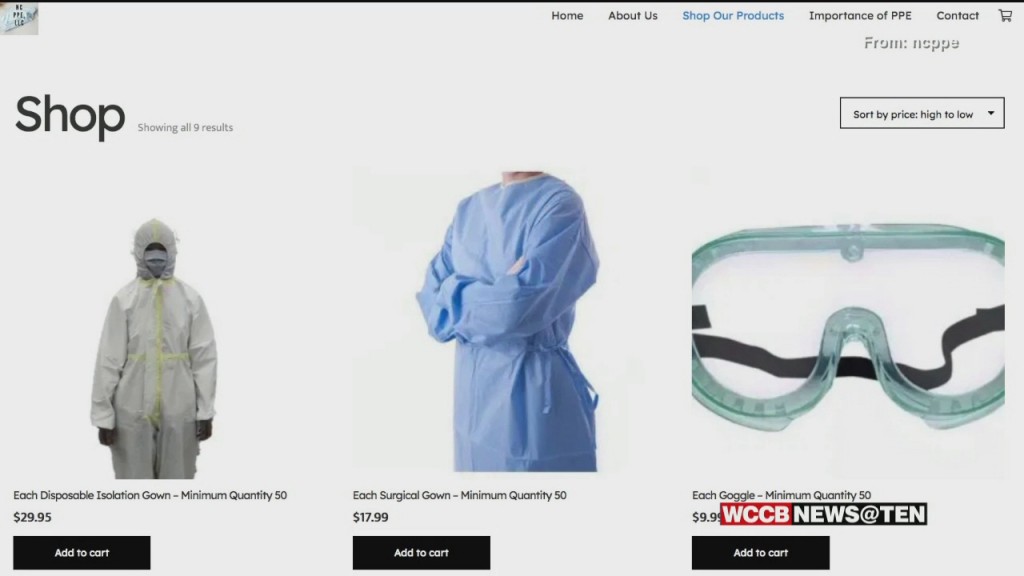
LabCorp, a North Carolina-based global life sciences company, has announced that it has received an Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) for a COVID-19 self-collection kit.